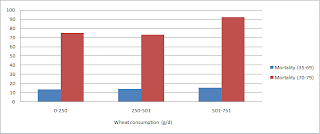
In this post on the China Study II data we have seen that wheat apparently displaces dietary fat a lot, primarily fat from animal sources. We have also seen in that post that wheat is strongly and positively associated with mortality in both the 35-69 and 70-79 age ranges, whereas dietary fat is strongly and negatively associated with mortality in those ranges.
This opens the door for the hypothesis that wheat increased mortality in the China Study II sample mainly by displacing dietary fat, and not necessarily by being a primary cause of health problems. In fact, given the strong displacement effect discussed in the previous post, I thought that this hypothesis was quite compelling. I was partly wrong, as you’ll see below.
A counterintuitive hypothesis no doubt, given that wheat is unlikely to have been part of the diet of our Paleolithic ancestors, and thus the modern human digestive tract may be maladapted to it. Moreover, wheat’s main protein (gluten) is implicated in celiac disease, and wheat contains plant toxins such as wheat germ agglutinin.
Still, we cannot completely ignore this hypothesis because: (a) the data points in its general direction; and (b) wheat-based foods are found in way more than trivial amounts in the diets of populations that have relatively high longevity, such as the French.
Testing the hypothesis essentially amounts to testing the significance of two mediating effects; of fat as a mediator of the effects of wheat on mortality, in both the 35-69 and 70-79 age ranges. There are two main approaches for doing this. One is the classic test discussed by Baron & Kenny (1986). The other is the modern test discussed by Preacher & Hayes (2004), and extended by Hayes & Preacher (2010) for nonlinear relationships.
I tested the meditating effects using both approaches, including the nonlinear variation. I used the software WarpPLS for this; the results below are from WarpPLS outputs. Other analyses of the China Study data using WarpPLS can be found here (calorie restriction and longevity), and here (wheat, rice, and cardiovascular disease). For yet other studies, click here.
The graphs below show the path coefficients and chance probabilities of two models. The one at the top-left suggests that wheat flour consumption seems to be associated with a statistically significant increase in mortality in the 70-79 age range (beta=0.23; P=0.04). The effect in the 35-69 age range is almost statistically significant (beta=0.22; P=0.09); the likelihood that it is due to chance is 9 percent (this is the meaning of the P=0.09=9/100=9%).
The graph at the bottom-right suggests that the variable “FatCal”, which is the percentage of calories coming from dietary fat, is indeed a significant mediator of the relationships above between wheat and mortality, in both ranges. But “FatCal” is only a partial mediator.
The reason why “FatCal” is not a “perfect” mediator is that the direct effects of wheat on mortality in both ranges are still relatively strong after “FatCal” is added to the model (i.e., controlled for). In fact, the effects of wheat on mortality don’t change that much with the introduction of the variable “FatCal”.
This analysis suggests that, in the China Study II sample, one of wheat’s main sins might indeed have been to displace dietary fat from animal sources. Wheat consumption is strongly and negatively associated with dietary fat (beta=-0.37; P<0.01), and dietary fat is relatively strongly and negatively associated with mortality in both ranges (more in the 70-79 age range).
Why is dietary fat more protective in the 70-79 than in the 35-69 age range, with the latter effect only being significant at the P=0.10 level (a 10 percent chance probability)? My interpretation is that, as with almost any dietary habit, it takes years for a chronically low fat diet to lead to problems. See graph below; fat was not a huge contributor to the total calorie intake in this sample.
The analysis suggests that wheat also caused problems via other paths. What are them? We can’t say for sure based on this dataset. Perhaps the paths involve lectins and/or gluten. One way or another, the relationship is complex. As you can see from the graph below, the relationship between wheat consumption and mortality is nonlinear for the 70-79 age range, most likely due to confounding factors. The effect size is small for the 35-69 age range, even though it looks linear or quasi-linear in that range.
As you might recall from this post, rice does NOT displace dietary fat, and it seems to be associated with increased longevity. Carbohydrate content per se does not appear to be the problem here. Both rice and wheat foods are rich in them, and have a high glycemic index. Wheat products tend to have a higher glycemic load though.
And why is dietary fat so important as to be significantly associated with increased longevity? This is not a trivial question, because if too much of that fat is stored as body fat it will actually decrease longevity. Dietary fat is very calorie-dense, and can be easily stored as body fat.
Dietary fat is important for various reasons, and probably some that we don’t know about yet. It leads to the formation of body fat, which is not only found in adipocytes or used only as a store of energy. Fat is a key component of a number of important tissues, including 60 percent of our brain. Since fat in the human body undergoes constant turnover, more in some areas than others, lack of dietary fat may compromise the proper functioning of various organs.
Without dietary fat, the very important fat-soluble vitamins (A, D, E and K) cannot be properly absorbed. Taking these vitamins in supplemental form will not work if you don’t consume fat as well. A very low fat diet is almost by definition a diet deficient in fat-soluble vitamins, even if those vitamins are consumed in large amounts via supplements.
Moreover, animals store fat-soluble vitamins in their body fat (as well as in organs), so we get these vitamins in one of their most natural and potent forms when we consume animal fat. Consuming copious amounts of olive and/or coconut oil will not have just the same effect.
References
Baron, R. M., & Kenny, D. A. (1986). The moderator–mediator variable distinction in social psychological research: Conceptual, strategic, and statistical considerations. Journal of Personality & Social Psychology, 51(6), 1173-1182.
Preacher, K.J., & Hayes, A.F. (2004). SPSS and SAS procedures for estimating indirect effects in simple mediation models. Behavior Research Methods, Instruments, & Computers, 36 (4), 717-731.
Hayes, A. F., & Preacher, K. J. (2010). Quantifying and testing indirect effects in simple mediation models when the constituent paths are nonlinear. Multivariate Behavioral Research, 45(4), 627-660.
Monday, February 21, 2011
Sunday, February 6, 2011
Is working standing up too expensive? It could cost you as little as $10
Spending too much time sitting down is clearly unnatural, particularly if you sit down on very comfortable chairs. Sitting down per se is probably natural, given the human anatomy, but not sitting down for hours in the same position. Also, comfortable furniture is an apparently benign Neolithic invention, but over several years it may stealthily contributed to the metabolic syndrome and the diseases of civilization.
Getting an elevated workstation may be a bit expensive. At work, you may have to go through a bit of a battle with your employer to get it (unless you are "teh boz"), only to find out that having to work standing up all the time is not what you really wanted. That may not be very natural either. So what is one to do? One possible solution is to buy a small foldable plastic table (or chair) like the one on the figure below, which may cost you less than $10, and put it on your work desk. I have been doing this for quite a while now, and it works fine for me.
The photo above shows a laptop computer. Nevertheless, you can use this table-over-table approach with a desktop computer as well. And you still keep the space under the foldable table, which you can use to place other items. With a desktop computer this approach would probably require two foldable tables to elevate the screen, keyboard, and mouse. This approach also works for reading documents and writing with a pen or pencil; just put a thick sheet of paper on the foldable table to make a flat surface (if the foldable table’s surface is not flat already). And you don’t have to be standing up all the time; you can sit down as well after removing the foldable table. It takes me about 5 seconds to do or undo this setup.
When you sit down, you may want to consider using a pillow like the one on the photo to force yourself to sit upright. (You can use it as shown, or place the pillow flat on the chair and sit on its edge.) Sitting on a very comfy chair with back support prevents you from using the various abdominal and back muscles needed to maintain posture. As a result, you may find yourself unusually prone to low back injuries and suffering from “mysterious” abdominal discomfort. You will also very likely decrease your nonexercise activity thermogenesis (NEAT), which is a major calorie expenditure regulator.
With posture stabilization muscles, as with almost everything else in the human body, the reality is this: if you don’t use them, you lose them.
Getting an elevated workstation may be a bit expensive. At work, you may have to go through a bit of a battle with your employer to get it (unless you are "teh boz"), only to find out that having to work standing up all the time is not what you really wanted. That may not be very natural either. So what is one to do? One possible solution is to buy a small foldable plastic table (or chair) like the one on the figure below, which may cost you less than $10, and put it on your work desk. I have been doing this for quite a while now, and it works fine for me.
The photo above shows a laptop computer. Nevertheless, you can use this table-over-table approach with a desktop computer as well. And you still keep the space under the foldable table, which you can use to place other items. With a desktop computer this approach would probably require two foldable tables to elevate the screen, keyboard, and mouse. This approach also works for reading documents and writing with a pen or pencil; just put a thick sheet of paper on the foldable table to make a flat surface (if the foldable table’s surface is not flat already). And you don’t have to be standing up all the time; you can sit down as well after removing the foldable table. It takes me about 5 seconds to do or undo this setup.
When you sit down, you may want to consider using a pillow like the one on the photo to force yourself to sit upright. (You can use it as shown, or place the pillow flat on the chair and sit on its edge.) Sitting on a very comfy chair with back support prevents you from using the various abdominal and back muscles needed to maintain posture. As a result, you may find yourself unusually prone to low back injuries and suffering from “mysterious” abdominal discomfort. You will also very likely decrease your nonexercise activity thermogenesis (NEAT), which is a major calorie expenditure regulator.
With posture stabilization muscles, as with almost everything else in the human body, the reality is this: if you don’t use them, you lose them.
Subscribe to:
Posts (Atom)